|
What types of intermolecular forces are present in HCl? In this article, you will learn everything you need to know about the intermolecular forces in HCl. Hey Readers!!! Welcome to another fresh article on techiescientist. 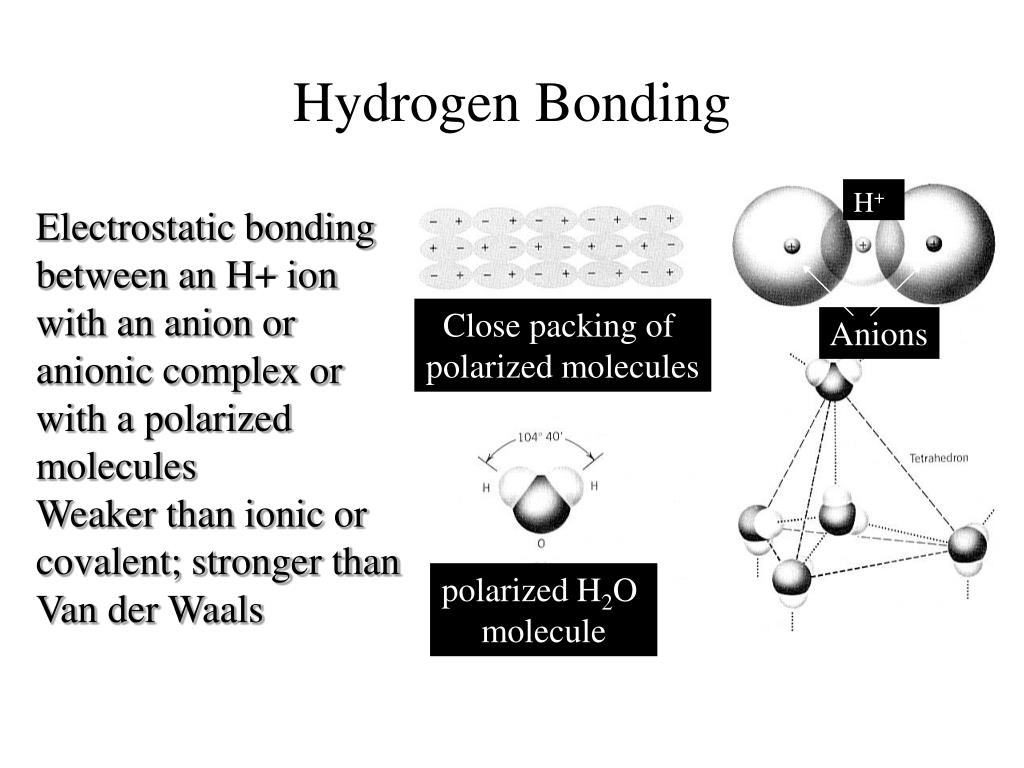
It is used in the production of a number of inorganic compounds, in the pickling of steel, in pH control and neutralization reactions, etc. It is also found as a component of gastric acid in the stomach of humans as well as some other animals. In the industries, hydrochloric acid is prepared by reacting hydrogen chloride with water. 
it contains one atom of hydrogen and one atom of chlorine. It is denoted by the chemical formula HCl i.e. Are intermolecular forces weaker than the intramolecular forces?Īnswer: Yes, intermolecular forces are weaker than the intramolecular forces because the attraction between the same molecule that helps to hold the atom together in the same molecular species is stronger than the attraction that helps to hold between two different molecular species.Hydrochloric acid is a colorless, pungent-smelling liquid. Besides of temperature, pressure, kinetic energy have an impact of intermolecular forces between the molecules. Which factors affect the intermolecular forces?Īnswer: The strength of attraction between the molecules is the most important determining factor of intermolecular forces. They are generated due to the attraction between two oppositely charged species. To know more please follow: Properties of Peptide bond: Detailed Fact and Comparative Analysis Frequently Asked Questions (FAQ) What causes the intermolecular forces between the molecules?Īnswer: Most of the intermolecular forces are electrostatic in nature. Hydrogen bonding, dipole-dipole interaction, dipole-induced dipole interaction are stronger than the London dispersion force. 
The constant motion of the atoms or molecules can cause an instantaneous dipole due to the unsymmetrical distortion of the electron cloud around the nucleus. This dispersion force is generated when the electrons from two adjacent atoms orient in such way that makes the atom into a temporary dipole. London dispersion force is a type of very weak intermolecular force between two molecules when they are in close proximity with each other. London Dispersion Force is the interaction between one induced dipole and instantaneous dipole. It is stronger than the dipole-dipole interaction. The charge of the ion distorts the electron cloud of the nonpolar molecule and as a result the molecule becomes partially charged.Īmount of charge and charge density of ion increases the strength of ion-induced dipole interaction. Thus one negatively polarized and a positively polarized end will be created in that molecule after the induction by the ion. Ion is a charged species and it can induce (disturbing the arrangement of the inner electrons) any nonpolar and neutral molecule. 
Image Credit: Wikimedia Commons Ion-Induced Dipole Interaction Hydrogen bond is comparatively stronger than Vander waals force but weaker than covalent bonding. The bond dissociation energy or bond energy of a hydrogen bond depends on the nature of acceptor, donor atoms, geometry and environment. The most familiar hydrogen bond acceptor and donor is Oxygen, nitrogen, fluorine having greater electronegativity.īoth type of hydrogen bonding is known in chemistry, that is intermolecular and intramolecular hydrogen bonding. Hydrogen bond is basically an electrostatic force of attraction acts between one hydrogen atom, covalently bonded with an electronegative atom, with another electronegative atom known as hydrogen bond acceptor from same or different molecule. The different types of intermolecular forces (interaction between two different or two same molecules) are written below. In this article “intermolecular forces examples”, the different types and examples of the intermolecular forces are explained briefly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
